2023 Anthony J. MacKay Student Paper Contest Winner

Every year, CRPA’s Student and Young Professionals Committee organizes the Anthony J. MacKay Student Paper Contest. The contest is open to all students enrolled full-time in a Canadian college or university program related to radiation sciences. From among the submissions, three finalists are selected and given the opportunity to present their work in a plenary session at the conference. The presentations are judged, and the winner is announced during the awards banquet.
The winner of this year’s contest was Bryce Nelson, PhD candidate in oncology, University of Alberta. You can read his submission below.
You can also read the submissions from the other two finalists in the article “2023 Anthony J. MacKay Student Paper Contest,” which was published in the Bulletin on May 17, 2023.
High-Yield Cyclotron Production of Pb-203 Using a Sealed Tl-205 Solid Target
Bryce Nelson, PhD candidate in oncology, University of Alberta
Coauthors
- John Wilson, Jan Andersson, and Frank Wuest, University of Alberta
- Jonathan Doupe, Alberta Health Services
- Michael Schultz, University of Iowa
Introduction
Nuclear medicine theranostics involves labelling a biological targeting vector first with a radionuclide (radioactive atom) for diagnostic imaging, followed by a particle-emitting radionuclide for targeted radionuclide therapy. Lead-212 (Pb-212, t1/2 = 10.6 h) is a particularly attractive therapeutic radionuclide due to its payload of one alpha and two beta− particles in its decay chain and the rapid decay of its progeny to stable Pb-208.
A recent clinical trial (Delpassand et al., 2022) using [Pb-212]Pb-DOTAMTATE to treat metastatic neuroendocrine tumours resulted in an 80% overall patient response rate, significantly exceeding standard-of-care treatments. However, diagnostic scans to track Pb-212 therapy were performed with conventional fluorine-18 and gallium-68 radiopharmaceuticals. This is suboptimal, as dissimilar chemistries between the diagnostic and therapeutic radionuclides could alter the structural conformation of the radiopharmaceutical and result in different biodistribution between the diagnostic and therapeutic compounds. The differences between the biodistribution observed in diagnostic imaging scans and subsequent biodistribution of the therapeutic radiopharmaceutical could, therefore, result in sub-optimal tumor dosing, or potentially unintended and destructive alpha-irradiation of healthy tissues.
As isotopes of an element are chemically indistinguishable, Pb-212 alpha therapy is ideally paired with the chemically identical lead-203 (Pb-203, t1/2 = 51.9 h) to provide diagnostic SPECT imaging using the 279 keV (81%) gamma-photons emitted during Pb-203 decay. However, worldwide supply of Pb-203 is extremely limited since cyclotron Pb-203 production requires irradiating highly toxic thallium (Tl) material.
Our objectives were to develop a high-yield Pb-203 cyclotron production route on the TR-24 Cyclotron (shown in Figure 1) in Edmonton’s Medical Isotope and Cyclotron Facility. We aimed to use use isotopically enriched Tl-205 target material and the Tl-205(p,3n) Pb-203 reaction as an alternative to lower energy production via the Tl-203(p,n) Pb-203 reaction. A robust cyclotron target and efficient chemical purification process must be designed to maximize Pb-203 yield and purity for research and clinical applications, while maintaining stringent radiation and chemical safety given the significant hazards presented to the operators and cyclotron facility.

Figure 1: TR-24 cyclotron in Edmonton’s Medical Isotope and Cyclotron Facility.
Methods
Our entire process was designed around preserving radiation and chemical safety. To reduce Tl contamination risk, we employed our patent-pending sealed cyclotron target design (shown in Figure 2) that is used to produce other radionuclides.
Tl-205 metal (99.9% isotopic enrichment) was pressed using a hardened stainless-steel die. High purity aluminum (Al) target discs (99.999%) were machined with a central depression and annulus groove.
A Tl-205 pellet was placed into the central depression of the Al disc, and indium wire was laid in the annulus. An aluminum foil cover was then pressed on, cold welding the cover to the disc via the indium with an airtight bond.
Targets were irradiated at 23.3 MeV for up to 516 minutes on a TR-24 cyclotron at proton currents up to 60 microamps to produce Pb-203 via the Tl-205(p,3n)Pb-203 nuclear reaction. Following a period of at least 12 hours to allow decay of Pb-204m (t1/2 = 67 min, 899 keV [99%]), the target was removed and Tl-205 was dissolved in HNO3.
A NEPTIS Mosaic-LC synthesis unit performed automated separation using Eichrom Pb resin, and Pb-203 was eluted with hydrochloric acid or ammonium acetate. The waste solution was diverted to a vial for subsequent Tl-205 recovery in an electrolytic cell.
Pb-203 product radionuclidic and elemental purity were assessed by high-purity germanium (HPGe) gamma spectroscopy and inductively coupled plasma optical emission spectroscopy (ICP-OES), respectively. Radiolabelling and blood serum stability studies were performed with PSC, TCMC, and DOTA chelators, and to stably sequester 203Pb for attachment to a biological targeting vector. Pb-203 chelation incorporation was verified by radio-thin layer chromatography (radio-TLC) analysis.
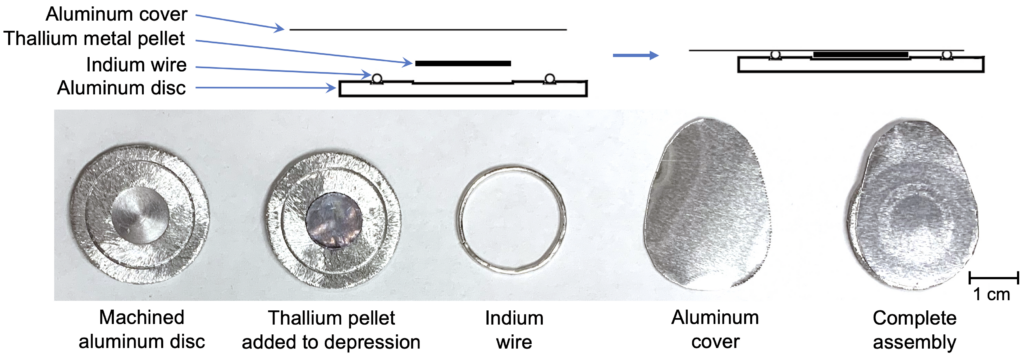
Figure 2: Sealed solid target assembly designed to encapsulate toxic thallium metal for cyclotron irradiation.
Results
Cyclotron irradiations were performed at a 60 microamps proton beam current and 23.3 MeV energy without any observable target degradation or thallium leakage. Automated purification took less than 4 hours, yielding greater than 85% decay-corrected Pb-203 with a radionuclidic purity of over 99.9% (gamma spectrum shown in Figure 3). Purified Pb-203 yields up to 12 GBq were attained, and Pb-203 was successfully chelated and exhibited over 99% incorporation after 120 hours in human serum for each of the PSC, TCMC, and DOTA chelators.
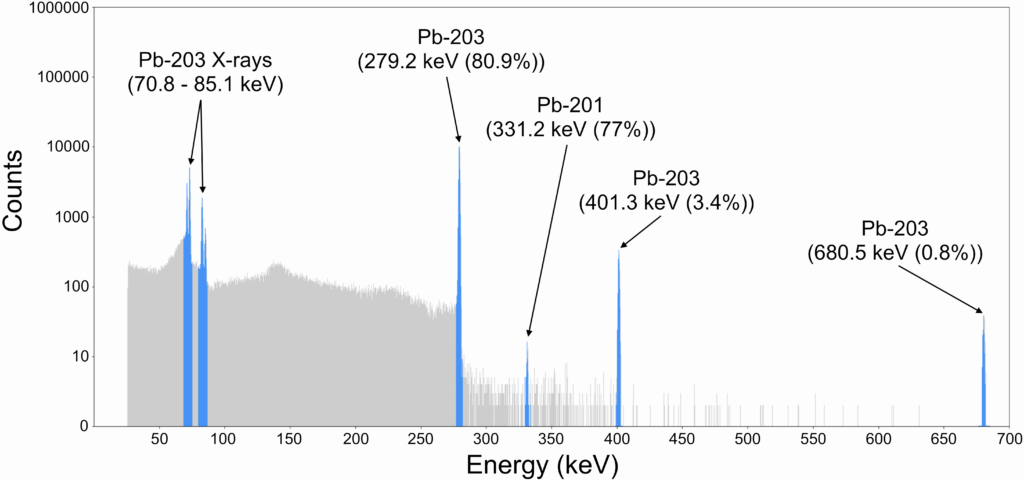
Figure 3: HPGe gamma spectrum of purified Pb-203 product.
Radiation safety implications: In over 100 production runs, there were no cyclotron target station or processing equipment contamination incidents.
Targets were irradiated during the afternoon and removed the following morning to permit decay of short-lived impurities. By allowing over 12 hours (over 10 Pb-204m half-lives) prior to target removal, operator dose was minimized.
Utilizing high purity Al target components nearly eliminated long-lived activation products, minimizing operator extremity dose when performing target recycling. When combined with a target retrieval shielding cart, a custom-designed lead-shielded processing castle, and an efficient automated process, the operator radiation dose per full production run is less than 10 µSv (measured by electronic personal dosimeter).
Conclusion
Our recently published high-yield Pb-203 production process significantly enhances Pb-203 production capabilities to meet the rapidly growing worldwide preclinical and clinical demand for Pb-203 radiopharmaceuticals alongside Pb-212 alpha particle therapy (Nelson, B. J. B. et al., 2023).
Since January 2022, we have successfully shipped over 70 batches of Pb-203 to customers and collaborators in 13 locations across 5 countries for research and diagnostic SPECT imaging clinical trials of metastatic melanoma and neuroendocrine tumours. Although we anticipate continually increasing demand, we are confident that our robust process design will maintain operator radiation dose at a small fraction of the permitted annual limit.
 Bryce Nelson
Bryce Nelson
PhD candidate in oncology, University of Alberta
Bryce obtained his BSc in chemical engineering from the University of Alberta (U of A) and is currently a PhD candidate in oncology at Edmonton’s Medical Isotope and Cyclotron Facility and the Cross Cancer Institute. Bryce is working under the supervision of Dr. Frank Wuest and Dr. Jan Andersson. His research involves developing cyclotron radionuclide production and automated purification processes, as well as applying radionuclides for targeted cancer diagnosis and therapy. Bryce is currently the president of the U of A Oncology Graduate Students’ Association and co-president of the U of A Bench to Bedside Students’ Association.
Do you want to read more articles like this?
The Bulletin is published by the Canadian Radiation Protection Association (CRPA). It’s a must-read publication for radiation protection professionals in Canada. The editorial content delivers the insights, information, advice, and valuable solutions that radiation protection professionals need to stay at the forefront of their profession.
Sign up today and we’ll send you an email each time a new edition goes live. In between issues, check back often for updates and new articles.
Don’t miss an issue. Subscribe now!
Subscribe

 Bryce Nelson
Bryce Nelson
